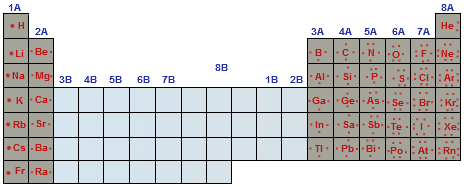
In single electron atoms, all energy levels with the same principle quantum number are degenerate, and have the same energy. For large n, the energy increases so much that the electron can easily escape from the atom. The n = 1 orbital has the lowest possible energy in the atom. In atoms with a single electron the energy of an orbital is determined exclusively by the principle quantum number n. For lanthanides and actinides, the number of valence electrons ranges from 3 to 16 ( ns, ( n−2)f and ( n−1)d orbitals).Īll other non-valence electrons for an atom of that element are considered core electrons.Ī more complex explanation of the difference between core and valence electrons can be described with atomic orbital theory. 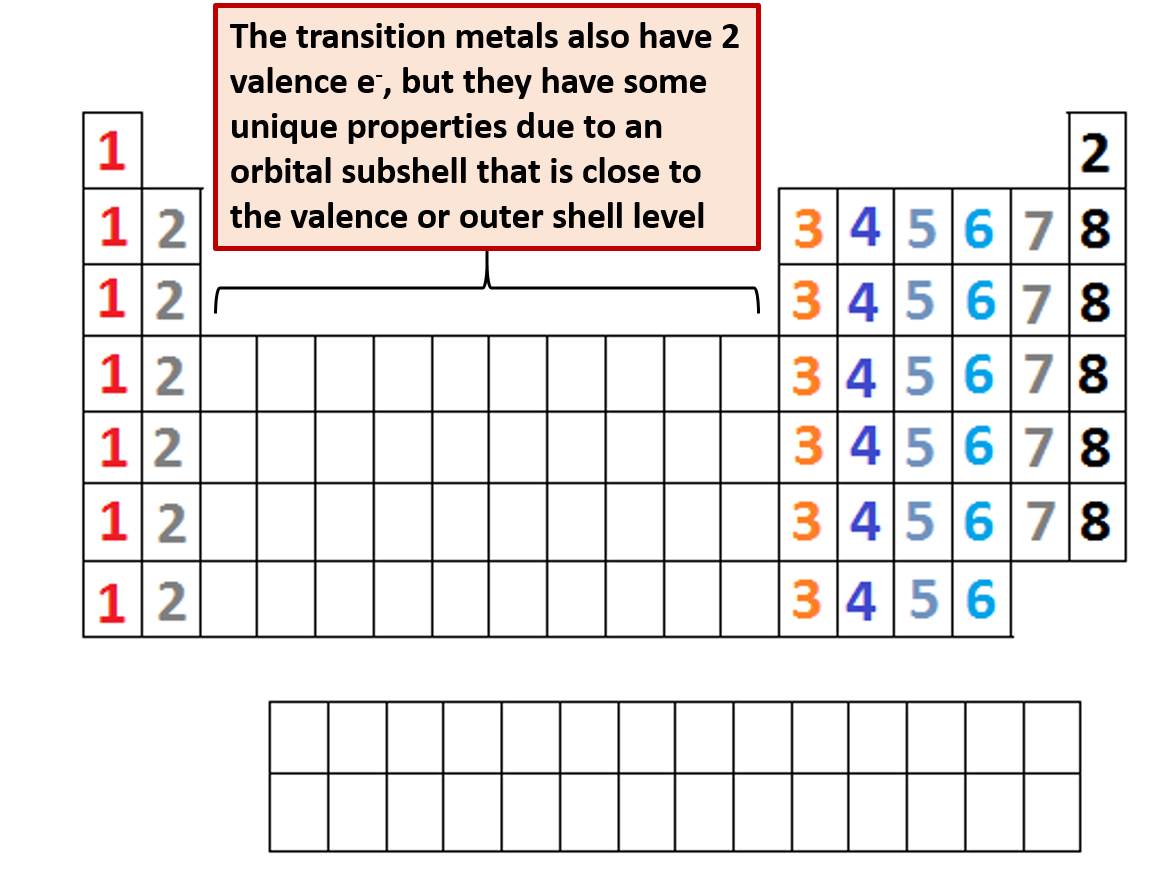
For transition metals, the number of valence electrons ranges from 3 to 12 ( ns and ( n−1)d orbitals).For main-group elements, the number of valence electrons ranges from 1 to 8 ( ns and np orbitals).The number of valence electrons of an element can be determined by the periodic table group of the element (see valence electron): Therefore, unlike valence electrons, core electrons play a secondary role in chemical bonding and reactions by screening the positive charge of the atomic nucleus from the valence electrons. Core electrons are tightly bound to the nucleus. The nucleus and the core electrons of an atom form the atomic core. This is strictly true for all elements in the s and p blocks.Core electrons are the electrons in an atom that are not valence electrons and do not participate in chemical bonding. Elements in each column have the same valence shell electron configurations, and the elements have some similar chemical properties. The same concept applies to the other columns of the periodic table. The organization of electrons in atoms explains not only the shape of the periodic table, but also the fact that elements in the same column of the periodic table have similar chemistry. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry- and they do. 
They all have a similar electron configuration in their valence shells: a single s electron. The second column on the right has the different corresponding electron configurations for the specified element in the rows underneath. The first column on the left has various elements in the rows underneath. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Table with two columns and 6 rows. 
For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. If we look at just the valence shell's electron configuration, we find that in each column, the valence shell's electron configuration is the same. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. S block is blue, d block is green, p block is red and f block is orange. Empty periodic table, separated into 4 different colored blocks. The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
